13.4: Ways of Expressing Concentration - Chemistry LibreTexts
4.8 (151) · € 14.50 · En Stock
Different units are used to express the concentrations of a solution depending on the application. The concentration of a solution is the quantity of solute in a given quantity of solution. It can be …
Different units are used to express the concentrations of a solution depending on the application. The concentration of a solution is the quantity of solute in a given quantity of solution. It can be expressed in several ways.

13.4: Solution Concentration - Chemistry LibreTexts
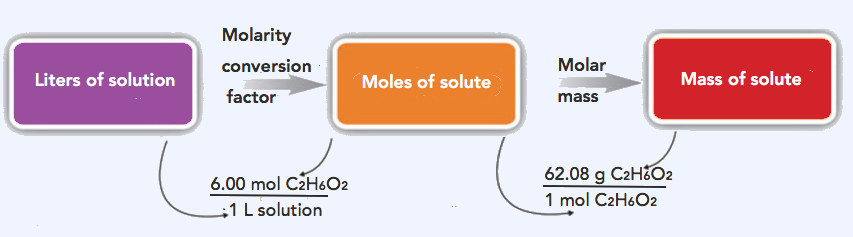
9.6: Units of Concentration - Chemistry LibreTexts

15.2 pH and pOH – Chemistry Fundamentals

8.6 Other Units for Solution Concentrations – Chemistry Fundamentals

Chapter 15.4: Non-equilibrium Conditions - Chemistry LibreTexts
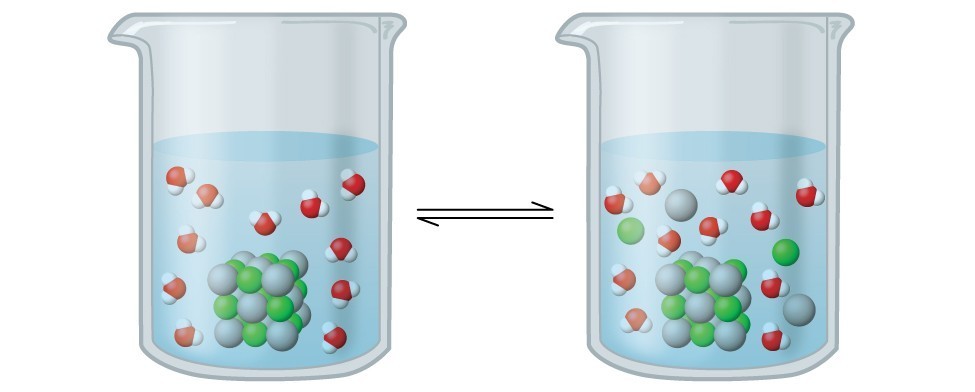
16.1 Precipitation and Dissolution – Chemistry Fundamentals

12.1 Expressing Concentration The Amount of Solute in the solvent - ppt download

The Beer-Lambert Law - Chemistry LibreTexts

The Beer-Lambert Law - Chemistry LibreTexts

antibiotics are overprescribed and mechanisms of horizontal gene trans
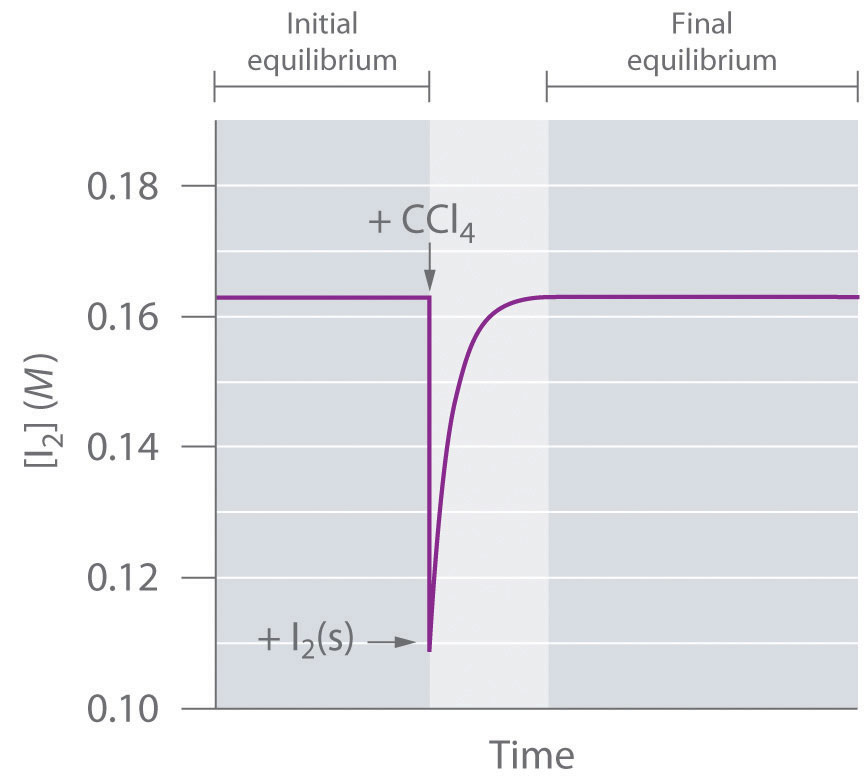
Chapter 14.5: Factors That Affect Equilibrium - Chemistry LibreTexts

Indoor acids and bases - Nazaroff - 2020 - Indoor Air - Wiley Online Library
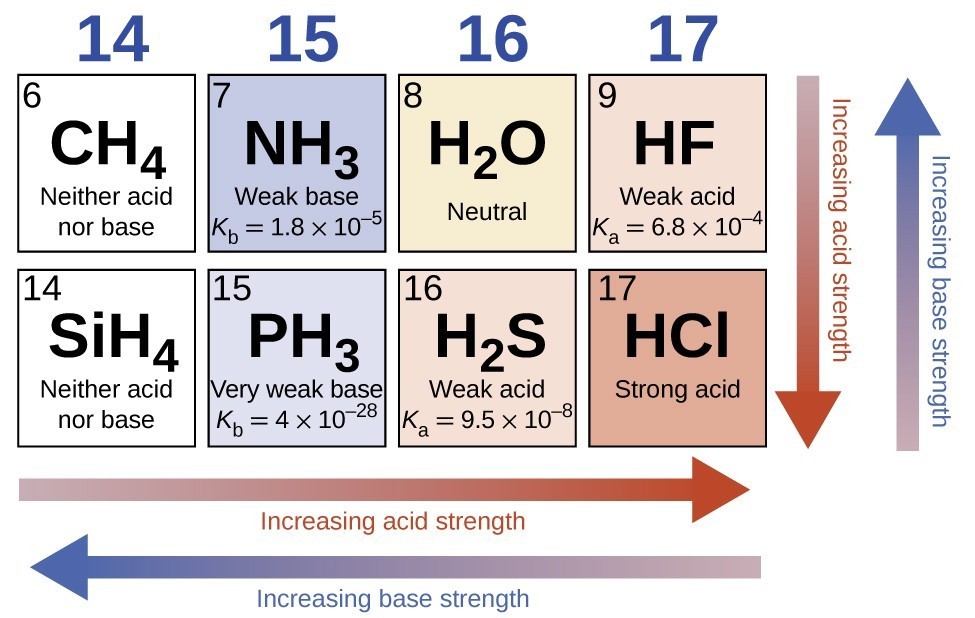
15.3 Relative Strengths of Acids and Bases – Chemistry Fundamentals